Non-invasive Diabetes Tech: The Never-ending Dream
The invention of a "noninvasive" device that can measure blood glucose for people with diabetes with no need to poke the skin and draw blood has been the dream for decades.
After all, why wouldn't people with diabetes (PWDs) flock to a skin patch that can detect blood sugar levels through sweat, or a wrist band that uses radio frequency technology to continuously beam glucose data directly to an app?
Numerous companies are pushing forward in this noninvasive continuous glucose monitoring (CGM) space, even in the midst of a pandemic.
To date, it's been mostly hype versus hope, as attempts to create those products have fallen flat.
The Diabetes Technology Society (DTS) published a scientific analysis on this topic in October 2021, noting "the amount of interest in seeing the development of an accurate [noninvasive glucose sensor] and the amount of hyperbole by companies promising an accurate [product] both far outstrip the amount of publicly available data actually generated by these potential products."
Yet, diabetes technology experts still believe potential exists for noninvasive devices to make it big, and industry analysts are predicting a booming market in the next 5 years.
How does it work?
Analysts note that there are systems under development for both home use and in-clinic and hospital settings. The former are wearables, and the latter will be non-wearable or tabletop systems.
They segment the systems under development by the type of technology used to take blood glucose readings — mainly, different types of spectroscopy, a technique that identifies chemicals based on the interaction of molecules with electromagnetic radiation.
Spectroscopy, which uses lasers that don’t pierce the skin, has been under study for decades. Researchers at MIT and elsewhere are finding that when used properly, it can produce highly accurate continuous data on blood glucose levels.
In a June 2021 academic review article, the DTS — led by Dr. David Klonoff of the University of California, San Francisco and medical director of the Diabetes Research Institute at Mills-Peninsula Medical Center — highlighted the many barriers that exist, but still predicted that noninvasive devices are poised for success in the coming years.
Notably, the article authors classified for the first time bloodless glucose monitoring products into three categories:
- Noninvasive optical glucose monitoring (NIO-GM): when a noninvasive measurement involves passing a type of radiation into a vascular region of the body
- Noninvasive fluid sampling (NIFS-GM): analyzes a fluid sample (tears, sweat, saliva, urine, etc.) that's collected without an invasive procedure
- Minimally invasive devices (MI-GM): involves insertion of a sensor into the subcutaneous tissue underneath the skin, such as existing continuous glucose monitoring technology that exists today
"Bloodless glucose monitoring products... are expected to become key components of novel wearable digital health tools for monitoring glucose concentrations in the diabetes market and the fitness market," the DTS authors wrote.
Who’s developing noninvasive CGM?
Let's take a look at some companies making progress. Keep in mind, there are ambitious new companies emerging in this space regularly, despite decades of others trying unsuccessfully.
Scanbo

At the big Consumer Electronics Show (CES) in early January 2022, an artificial intelligence company based in British Columbia named Scanbo gave a glimpse of its technology that would use a 60-second noninvasive finger measurement instead of a traditional blood drop required to measure glucose. The company has developed a prototype that combines a 3-lead ECG measurement and a Photoplethysmogram (PPG) used to detect blood volume. You just put your fingers on the flat white sensors and the system uses a set of algorithms to analyze and offer insight on glucose values.
Hagar Technology's GWave
Another new company making headlines is Hagar Technology, based in Israel, which received Food and Drug Administration (FDA) fast track designation last year after a series of investor fundraising. The company's developing what it calls the GWave, which is a sensor inserted into a "ceramic, lightweight, comfortable bracelet worn on the wrist" that uses radio frequencies to detect glucose levels. The device will be the size of a smartwatch and connect to a mobile app, enabling users to get glucose readings on their smartphones and share that data with their diabetes care team.
Clinical trials are in progress, according to the company's public comments.
SugarBEAT
SugarBEAT, from U.K.-based Nemaura Medical, is already approved for use in Europe. It's a small peel-and-place patch that sticks onto your skin for 24 hours before needing replacement. The adhesive-backed rectangular transmitter sends wireless readings to a companion smartphone app via Bluetooth every 5 minutes.
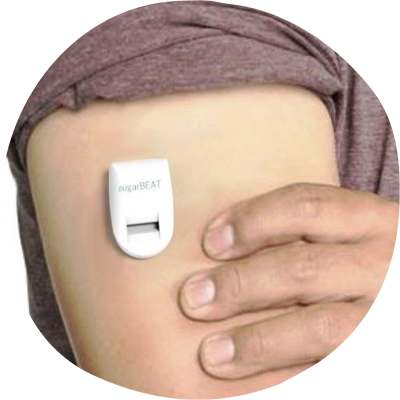
According to the company, it works by “passing a mild, nonperceptible electric current across the skin, (which) draws a small amount of selected molecules, such as glucose, into a patch placed on the skin. These molecules are drawn out of the interstitial fluid, which naturally sits just below the top layer of skin.”
Nemaura had originally submitted this to the FDA in mid-2019, but the company had to refile the following year with additional study data. Then, the pandemic began. The company tells DiabetesMine they hope to continue conversations with regulators as soon as possible in order to move forward, but there's no expected timeline available.
In the meantime, they're launching a nonregulated product called proBEAT in the United States, which is a professional CGM version developed for use in those with type 2 diabetes and others who don't need continuous glucose data. Their program incorporates the device into a meal replacement plan, originally developed by the Joslin Diabetes Center in Boston, Massachusetts, and overseen by healthcare providers.
Know Labs
Seattle, Washington-based Know Labs is developing two devices that employ Body-Radio Frequency Identification (Bio-RFID) technology, which uses radio waves to measure specific molecular signatures in the blood through the skin. Formerly known as Visualant, this tech company changed its name in 2018 and is developing both a wristband-style device as well as a finger-scanning device that eliminate the need to pierce the skin to get glucose readings.
- UBand is a wristband with a built-in biosensor designed to continuously monitor glucose levels. It won’t have a display screen, but instead, the readings will be sent directly to a smartphone app. They’re expecting it to have a battery life of 7 days before you would need to recharge it. This product is aimed at people with diabetes who are interested in a noninvasive CGM.
- KnowU is a device that sort of resembles a traditional fingerstick glucose meter in that it's small enough to fit into your pocket — but it doesn’t require test strips. Instead, you’d take the sensor off the base and scan it over your palm to generate readings that would then display on the base device itself or be beamed wirelessly to a smartphone app. This is aimed mainly at PWDs who aren’t interested in a full CGM, but want more of an easy on-the-go use option for glucose monitoring without the finger pokes.
Know Labs previously told DiabetesMine it hoped to begin the FDA pre-approval process in 2022.
DiaMonTech
Out of Germany, DiaMonTech has developed a system that uses molecular spectroscopy — the science of absorption of light by molecules — to detect glucose molecules through the skin. They're currently working on three versions:
- a phone-like "D-Pocket" handheld receiver that you press your finger on to get a glucose reading, which isn't expected before 2023 at the earliest
- a small "D-sensor" embedded into watches or fitness bands, which is anticipated for 2024 for later
- a more stationary, shoebox-sized device that would sit on a table and could be used in clinical settings, which is ready for use now

"Finger pricking is messy and inconvenient, and that’s why so many diabetics are not measuring enough to successfully manage their blood sugar," DiaMonTech CEO Thorsten Lubinski told DiabetesMine. "Noninvasive measurements lower this hurdle, as measuring your blood sugar is as easy as using the fingerprint sensor on your mobile phone."
Lubinski recognizes that others before have failed to develop noninvasive D-tech, but emphasizes that his company's technology is more accurate and focuses specifically on the glucose molecule itself (rather than other things).
Created by startup co-founder Dr. Werner Mäntele, this technology has shown in research from 2020 that it has comparable accuracy to the minimally invasive FreeStyle Libre Flash glucose monitor from Abbott Diabetes.
NovioSense
The Dutch startup known as NovioSense is working on a glucose sensor that is placed under the lower eyelid, from where it can wirelessly send glucose measurements directly to a smartphone.
The NovioSense device consists of a flexible metal coil just 2 centimeters long that contains nanosensors inside. The coil is covered by a protective layer of soft hydrogel, and it could measure constant changes in glucose levels from tear fluid using the same enzyme technology employed in conventional glucose test strips.
Clinical trial research published in late 2020 shows promising results for the technology and accuracy similar to the FreeStyle Libre, but there are few details available beyond that.
LifePlus
This Silicon Valley, California-based startup is developing a noninvasive wearable wristwatch called LifeLeaf. The company says it can detect blood glucose levels, blood pressure, heart rate, sleep apnea, and more by using sensors already on the market and an additional light sensor to enhance accuracy.
According to this demo presentation at DiabetesMine's D-Data ExChange forum in June 2020, it uses a "novel approach based on spectroscopic principles... to determine an accurate spectral signature for glucose."
Their phase 1 product is aimed at consumers and people with prediabetes, and phase 2 will be for type 2 PWDs and eventually also those with type 1, with high and low alerts and guidance.
The company has conducted clinical trials around the world, and at last count, was aiming for FDA clearance by June 2022.
Afon Technology

Out of Wales, a startup called Afon Technology is developing a sensor that would fit inside a smartwatch band to monitor glucose levels. Per the company's website, it will provide real-time data and alert PWDs to high and low sugars, as well as track other health data. The company is working on clinical trials outside the United States, with plans for a launch starting in mid-2022.
Afon shares feedback from Dr. Hans de Vries, medical director at diabetes research organization Profil in Germany and a principal investigator at the University of Amsterdam's Faculty of Medicine, who says: “We evaluated the Afon device under both hyper- and hypoglycemic conditions during the clinical trials and we were surprised and excited by the possibilities of this technology.”
Apple and Samsung
Both Apple and Samsung are rumored to be developing noninvasive CGM sensors of their own.
In fact, for years, many have confused the Apple Watch's capability to display Dexcom’s CGM data with the notion that Apple itself offers CGM technology. But that buzz may finally be coming true soon.
According to a January 2021 report, Apple may be working on their own glucose monitoring tech that would use an integrated optical glucose sensor. The report has some fascinating visuals on what the Apple Watch display could look like.
Samsung may have its sights on this tech, too. This January 2021 news report states:
“Samsung Electronics will be equipped with a blood glucose measurement function in the new smartwatch ‘Galaxy Watch 4’ [tentative name] to be introduced in the second half of this year. It is a no-blood sampling method that detects the level of glucose in the blood without blood collection using an optical sensor, and is expected to contribute to the health management of the general public as well as diabetics.”
There had been talk years back about a Samsung and Medtronic Diabetes partnership aimed at integrating glucose data into Android watches, but that relationship faded without any product materializing beyond prototypes.
There are numerous other small companies and universities currently working on noninvasive glucose monitoring technology, too.
Gone but not forgotten
DiabetesMine has been covering attempts at noninvasive diabetes tech since 2005, and a couple of the gadgets that captured headlines at the time remain legendary.
GlucoWatch
The first and best-known example is the infamous GlucoWatch. This product made it to market briefly in the early 2000s, but it ended up being totally unreliable and actually started burning some patients' skin while they were wearing it! It was later recalled by the FDA.
Contact lenses
Google and Novartis were once developing a contact lens that could use fluorescent light to monitor sugars through the eye, but that research was eventually scrapped because the company determined it wouldn't work. That hasn't stopped others from pursuing that same path in recent years.
C-8 MediSensors
Another notable name in noninvasive CGM tech for several years was C-8 MediSensors based in San Jose, California. This gadget promised to use light to identify and analyze glucose molecules under the skin via interstitial fluid, just like other traditional CGMs.
This company even obtained European CE Mark approval in 2012, but a launch never materialized and, eventually, the company went bankrupt a year later. Many of the C-8 scientists moved on to other companies like Apple and Google, before the company eventually rebranded and relaunched as C-Eight without any focus on noninvasive glucose monitoring.
What the skeptics say
Some industry experts who've watched this field for many years still insist that it will never happen.
"It's the easiest slide-deck pitch to make, but it never seems to go anywhere," said JDRF CEO Dr. Aaron Kowalski, who has been knee-deep in the world of emerging diabetes technology for decades. "There are major technology challenges that are so big, and you have to wonder if there is even a need anymore with what we have in diabetes technology today."
"Noninvasive still has a lot of challenges," said Dr. Barry Ginsberg, who runs Diabetes Technology Consultants in New Jersey and is considered a premier expert on noninvasive diabetes tech after analyzing this trend for more than a decade. He predicts the market will be dominated by products that are simply "less invasive" and smaller than what we have now, such as the Dexcom G7 model expected in 2022.
Semiretired industry consultant John L. Smith is another of the foremost experts on this front, author of the seminal paper “The Pursuit of Noninvasive Glucose: Hunting the Deceitful Turkey” article first published in 2006 and last updated in a 2020-seventh edition.
He writes that one of the most disturbing aspects in this field has been the “perennial” announcements by young companies that believe they’ve reached a solution for PWDs to no longer have to prick their fingers. Without exception, Smith says these announcements have been premature and are meant to generate hype, raising false hopes.
He estimates that it would take a minimum of 5 years and as much as $25 to $30 million in funding just to get a product to market, so it's no wonder that most have pretty much failed to date.
He points to the wearables technology trend in recent years as growing strong beyond diabetes, but notes that the economic impact of the COVID-19 pandemic will likely push out many wannabe noninvasive tech developers.
Diabetes industry analyst David Kliff, an insulin-using PWD himself and notorious devil’s advocate, has maintained longstanding skepticism over noninvasive tech during his 20-plus years of writing for Diabetic Investor.
“It’s one of the better scams I’ve ever seen in the diabetes world,” he told DiabetesMine. “It sounds so appealing with all kinds of 'Star Trek'-y elements. They sell the hype and there’s just enough science to back it up so it looks great. The mainstream media give it more play and people buy the dream.”
Kliff decries the millions of dollars that investors are tempted to "throw at this tech."
The base problem, he says, is not so much having an easier way to get a glucose reading, but knowing what to do with that reading in order to improve your health outcomes.
“I’ve contended from day one, that whether it’s invasive or not, if the person doesn’t understand the number, it doesn’t matter if it’s given to them by God. I’m more impressed with practical technology that works and you can trust. For any patient, that’s the bottom line.”
Nevertheless, researcher Smith writes about noninvasive glucose monitoring: “In spite of all the failures (and quite likely because I have been close to so many of them), it is still my fond wish that someday, somewhere, someone will find the solution to this intensely recalcitrant problem and realize the benefits for all people with diabetes worldwide.”


Comments
Thanks